The group « Neuroproteomics and Biomarkers » (C. Hirtz) aims to exploit the latest technological developments in proteomics for the discovery, validation and use of clinical biomarkers (proteins, PTMs, ARN, ADN, metabolites) in neurodegeneratives diseases.
Our team is contituted of 4 development engineer (Dr. J. Vialaret, E. Attina, L. Fichter et L. Tiers), 2 clinical technician (J. Kindermanns, N. Ginestet), 7 researchers (Pr. S. Lehmann, Pr. C. Hirtz, Dr. C. Delaby, Dr. X. Ayrignac, Dr. E. Moulis, Pr. P Labauge et Pr. D. Deville de Périère) and 5 PhD (P. Mohaupt, S. Coppens, ML. Pons, A. Amalric, S. Muccio).

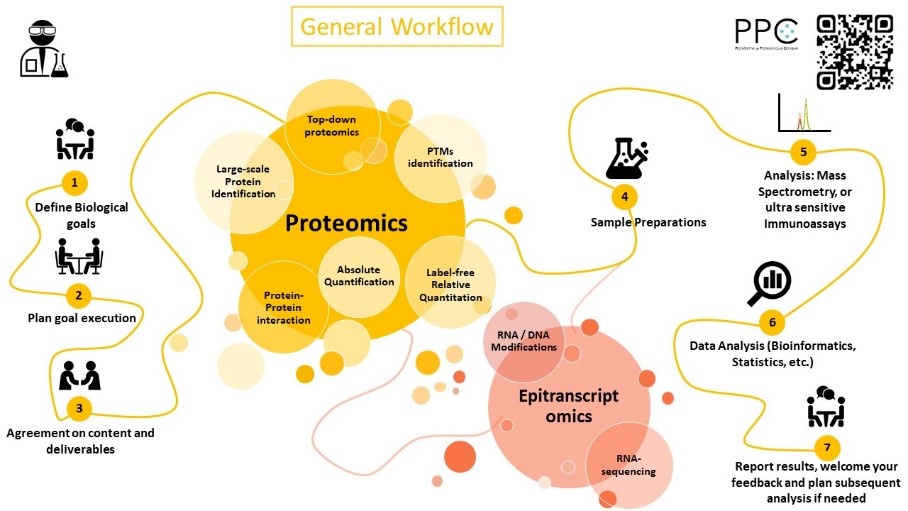
Our research are highly supported by a strong methodological development activities mainly dealing with prefractionation, preanalytical, innovative mass spectrometry approaches and clinical proteomics expertise.
To reach our goals, several high throughput technologies are available (targeted mass spectrometry, biomarkers discovery in complex matrices, ultra-sensitive and multiplexed immunoassays). For that purpose, we are currently equipped with last generation LC-MS/MS mass spectrometers (Q-TOF Impact 2 Bruker Daltonics et 3xTQ 8060 Shimadzu), a Maldi TOF (Axima Shimadzu), a high throughput sample preparation and fractionation robot (Bravo Assay Map Agilent) and ultra-sensitive and multiplexed immunoassays (SIMOA Quanterix HDX et Quickplex 120 MesoScale Discovery).


Highlights
- 2011 - First mass spectrometer triple quadrupole Agilent 6490
- 2012 - Collaboration contract with Agilent Technologies (Mass spectrometry development)
- 2014 - Collaboration contract with Bruker Technologies (Mass spectrometry development)
- 2018 – Member of the European Innovation Center Shimadzu (Mass spectrometry)
- 2019 - Collaboration with K. Tanaka (Prix Nobel Chimie 2002, Shimadzu) on Alzheimer’s biomarkers
Important publications 2011-2020
1- Lehmann S, Poinot P, Tiers L, Junot C, Becher F, Hirtz C. (2011) From "Clinical Proteomics" to "Clinical Chemistry Proteomics": considerations using quantitative mass-spectrometry as a model approach. Clin Chem Lab Med. Oct 8;50(2):235-42. (IF : 3.1)
2- Barthelemy N, Lehmann S, Hirtz C, Gabelle A, Sergeant N, Fenaille F, Vialaret J, Schraen S, Bros P, Tiers L, Delaby C, Junot C, Touchon J, Buee L, Becher F. Detection and Quantification of the Tau Protein and Its Isoforms in the CSF of Alzheimer's Disease Patients Using Mass Spectrometry. Alzheimer's & Dementia.10 (no. 4 sup): p. 147.(IF : 14.5)
3- Hirtz C, Vialaret J, Gabelle A, Nowak N, Dauvilliers Y and Lehmann S. From radioimmunoassay to mass spectrometry: a new method to quantify orexin-A (hypocretin-1) in cerebrospinal fluid. Scientific Reports. 2016 May 11;6: 25162. (IF:5.9)
4- Vialaret J, Wessels H, van Gool AJ, Lehmann S, Gabelle A, Wood J, Bern M, Paape R, Suckau D, Kruppa G and C Hirtz. Towards a routine application of Top-Down approaches for Label-Free discovery Workflows. Journal of Proteomics. 2018. Mar 20;175:12-26. (IF : 3.88).
5-Paterson RW, Gabelle A, Lucey BP, Barthélemy NR, Leckey CA, Hirtz C, Lehmann S, Sato C, Patterson BW, West T, Yarasheski K, Rohrer JD, Wildburger NC, Schott JM, Karch CM, Wray S, Miller TM, Elbert DL, Zetterberg H, Fox NC, Bateman RJ. SILK studies - capturing the turnover of proteins linked to neurodegenerative diseases. Nat Rev Neurol. 2019 Jul;15(7):419-427 (IF : 21.15)
Financements






Contact



















